By Genevieve Clapp, Second year, Medicine
Researchers at Bristol’s synthetic biology research centre BrisSynBio are working to design cells with minimal gene numbers using supercomputers.
Inside the nucleus of every living cell on this planet there lies a genome: a knot of genetic information that is entirely unique to you, so much so that even identical twins have small differences. Simply put, a genome is a chain of genes, which much like a zip, consist of pairs of interlocking molecules known as nitrogenous bases.
Humans have around 30 billion of these base pairs – frighteningly, this is also 30 billion places where the system can go wrong. A huge amount of time and resources has been dedicated to trying to uncover exactly what each gene does. Uncovering the patterns within the genome, has unlimited potential: allowing us to use genes like Lego bricks and construct organisms that can solve many of today’s problems. One such example would be designing bacteria that could convert waste material into feedstock or creating cancer drugs that target a specific gene.
| Yes we can-cer: shrinking tumors using the immune system
Here in Bristol, Head of the School of Biological Sciences Professor Claire Grierson has been supervising a team who are using computers to design genomes that are minuscule in comparison to normal bacterial DNA, let alone the human genome. The reasoning behind making a genome as small as possible is that as you remove genes, you begin to learn what they do. This is a more efficient version of what has been occurring in medicine for years, as often we work out the function of specific genes through the diseases that are caused in their absence.
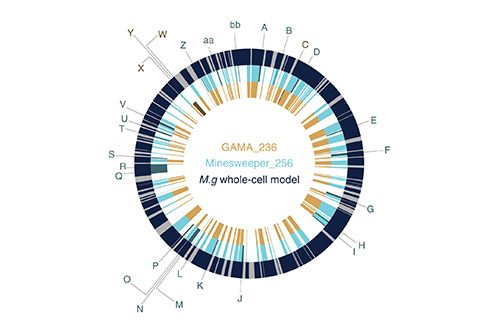
Many of us will have heard of the terms ‘in vivo’ and ‘in vitro’, however you may be less familiar with ‘in silico’. ‘In silico’ refers to testing a theory within a computerized model; Grierson’s team have adopted this method of research as it is far more efficient and predictable than using actual cells. They published their results in Nature Communications earlier in February.
Their program, Minesweeper, much like the game, searches for genes that could be deleted, then it runs a simulation to see if the cell will survive. The bacteria that they are using as a model is Mycoplasma genitalium– from its name you probably do not need me to tell you what kind of disease it causes. However, as well causing particularly nasty antibiotic resistant STI, it also has the smallest genome of any living organism (475 genes in total), hence it is perfect for experimentation.
| Lessons on tissue repair from fruit flies
Until recently the most its genome had been reduced was to 413 genes, but the Bristol team believe that if their simulation is correct, they can ‘knock out’ another 33 to 52 genes. For every gene that is knocked out we learn what is and is not essential to life. The aim is to eventually create a cell that is only capable of survival and nothing else: it will be a blank canvas upon which we can design completely new cells.
A design-simulate-test algorithms to design minimal genomes based on the whole-cell model of M. genitalium. @marucci_lucia @PR0FG @squishybinary @BristolBioSci @BrisBioDesignhttps://t.co/OHg3BssOeA
— Nature Communications (@NatureComms) February 14, 2020
One challenge that minesweeper and their other program, GAMA, have overcome is the fact that sometimes genes work in conjunction to produce a single trait. The danger with such genes is that if you delete a gene that works as part of a group, there may be no obvious effect and it could appear non-essential.
To avoid this, Minesweeper and GAMA do not begin by deleting single genes. Instead, they start by deleting a group of genes to see if they are working together. Once they have eliminated this possibility, they then begin to knock out the individual genes. Although projects like these have a long way to go before moving off the computer and into live organisms, it’s still a huge leap forward and something to be proud of.
Featured image: Flickr / ACJ1
Want to write about your research? Get in touch!