By Vilhelmiina Haavisto, SciTech Editor
Scientists from the University of Bristol’s School of Biochemistry have discovered how healing tissue protects itself from toxic, antibacterial compounds.
The body is quick to act when its tissues are damaged – immune cells are dispatched to the affected area within minutes, where they engulf and kill invading pathogens to prevent infection from taking root. They accomplish this feat by releasing toxic factors such as reactive oxygen species (ROS); while effective at killing bacteria, they can also disrupt the tissue’s own repair processes.
Bristol researchers have found a way to upregulate the cellular mechanisms that shield repairing tissues from the damaging effects of ROS. The study, published earlier this month in Current Biology, made use of fruit flies, a common model organism in life sciences research. However, these flies were translucent, meaning that their wound healing processes can be followed as they happen by tagging the molecules involved with fluorescent tags such as green fluorescent protein (GFP).
| The economics of antibiotic resistance
According to lead author Dr Helen Weavers, the group discovered a “network of protective pathways that shield tissues from inflammatory damage”, making healing tissues more [resilient]. Crucially, the researchers were able to show that the pathways can be activated ectopically; this means that fruit fly mutants that express the pathway components at levels different to those of non-mutant flies are created using gene editing. Ectopic expression was shown to further enhance the pathways’ protective properties; meanwhile, their inhibition caused “significant delays” in wound closure.
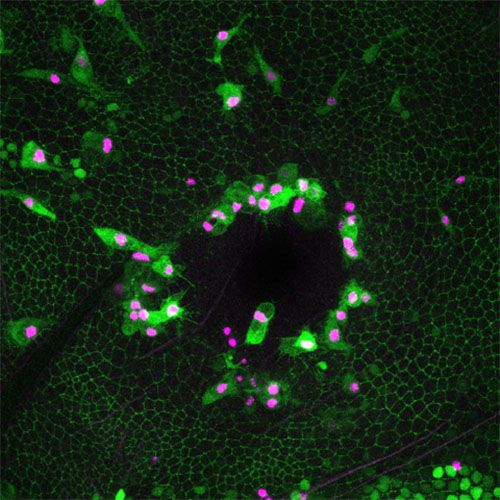
Damage and the resulting inflammation activate what are known as cytoprotective genes, whose function is to protect the cell from toxic factors such as ROS. The Gadd45 and dNrf2 resilience factors are products of such genes, and their ectopic expression before wounding was shown to increase the speed of the repair process and reduce DNA damage due to ultraviolet irradiation.
However, dNrf2 alone appears not to be enough to provide full stress-protection; the researchers speculate that several cytoprotective pathways may need to be activated to ensure maximum shielding. What’s more, long-term overexpression (production above normal levels) of dNrf2 also caused delays in wound closure, suggesting that more work needs to be done to fully understand what the study authors called the “cost” of increasing the productivity of these pathways.
| New synthetic vaccines developed using cloud computing
The work also uncovered some of the pathway’s evolutionary history. “[T]he protection machinery is activated by the same pathways that also initiate the inflammatory response”, noted Dr Weavers. This finding has led the researchers to believe that the protection machinery, including Gadd45 and dNrf2, has evolved as a “fail-safe mechanism” to protect tissues every time an inflammatory response is triggered.
With all the insights into cellular immunological processes, Dr Weavers says the team “hope[s] to develop ways to stimulate this protective machinery in patients prior to elective surgery”. Delays or complications in wound healing can have unwelcome consequences such as infection, often followed by a course of antibiotics, or further surgery, although this is relatively rare. Though this work in fruit flies is a long way from implementation in clinical settings, it is a step in the right direction.
Featured image: Flickr / Katya Schulz
Is this the way forward for wound healing? Tell us your thoughts!









