By Francesca Levi, Third Year, Biological Sciences
A new round of recruitment to the second phase of a clinical trial for the COVID-19 Oxford Vaccine (ChAdOx1 nCoV-19) has begun in Bristol. Jack Lennard and Evy Tang tell us about their experience being part of the COVID-19 trials.
Researchers at University Hospitals Bristol and Weston NHS Foundation Trust (UHBW), North Bristol NHS Trust (NBT) and the University of Bristol are currently recruiting up to 150 healthy Bristol volunteers over the age of 70. These volunteers will make up one of the groups in the study.
The vaccine, designed by researchers from the University of Oxford’s Jenner Institute and the Oxford Vaccine Group, is designed to protect healthy individuals against COVID-19.
The study will evaluate the vaccine’s safety and ability to generate a good immune response against the virus. Overall, phase II and III will be made up of 10,260 volunteers, including adults and children, in order to assess the response to the vaccine across different age groups.
The trial will see participants be randomly allocated to receive the ChAdOx1 nCoV-19 vaccine or a Men ACWY vaccine as a control. They will then do blood tests, nasal or throat swabs, and provide information about both COVID-19 related symptoms or any symptoms that could have resulted from the vaccine itself.
Currently recruiting up to 150 healthy Bristol volunteers over the age of 70
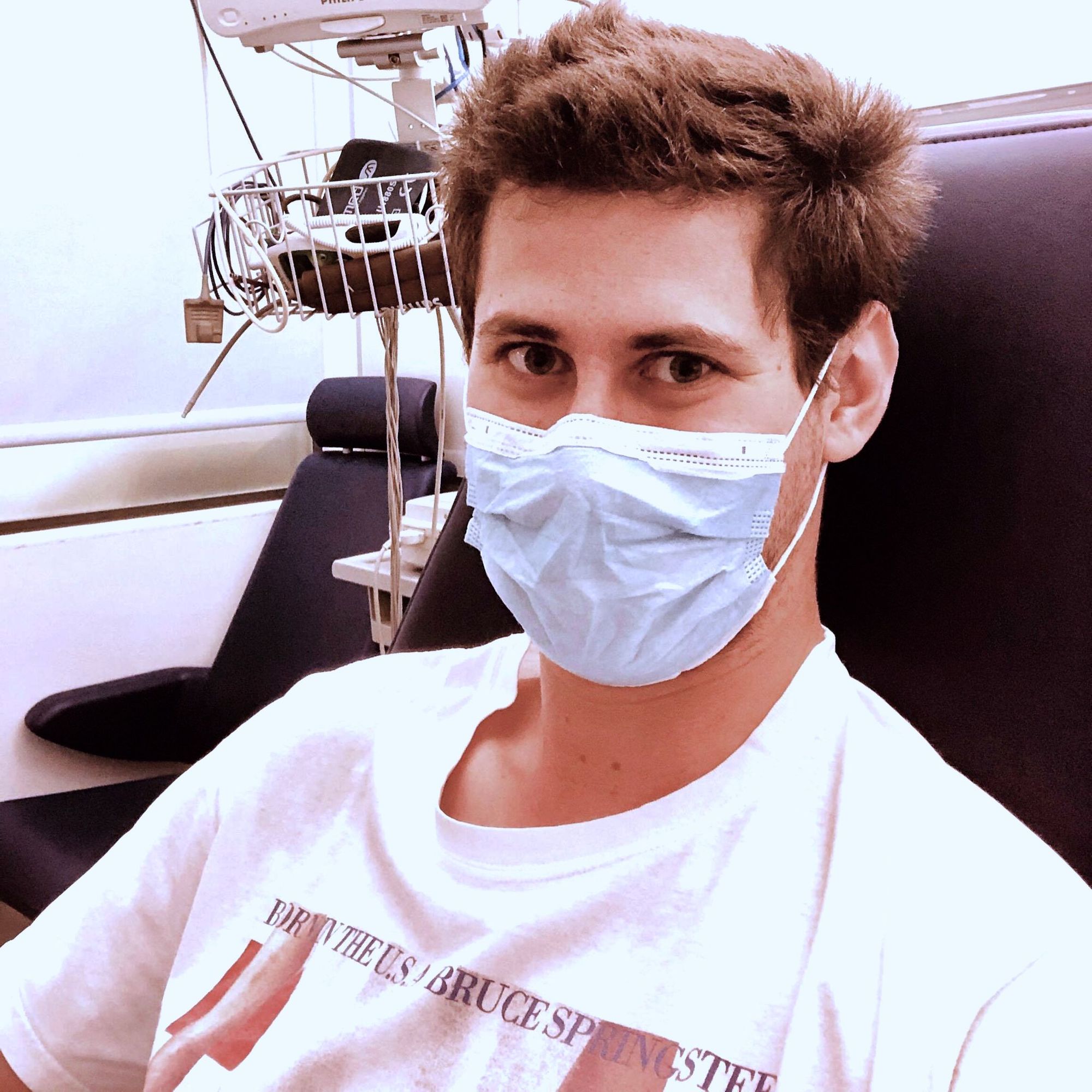
While Bristol is only now starting a new round of recruitment, in other locations in the United Kingdom the Oxford vaccine trials have been running since May. Here we speak with Jack Lennard, University of Oxford graduate and a participant of the ChAdOx1 nCoV-19 vaccine trial in London.
Which phase of the trial are you part of?
I’m in the phase II/III part of the trial. I applied for phase I, but their screening process took long so it filled up for phase I. By the time I got the green light, I was offered a place for phase II/III. I ended up getting the initial injection at the start of June, 90 days ago.
How did you first hear about the vaccine trials?
I was on Twitter. To be honest, I was doing the bare minimum of scrolling through social media and I saw that someone had tweeted information on signing up for the vaccine trials - so I thought 'that sounds fun'. They were running various studies across the country and one of them was the Southwest part of London. That’s where I am. So, I thought, why not?
Were there any specific requirements to take part in the study?
Health wise, you couldn’t do it if you had anything that would hinder your immune system. This means no history of cancer in the last five years, HIV, or hepatitis. They screen for that in person, even if you have already declared that you haven’t had it.
They also wanted to check that I didn’t have any ongoing serious medical conditions. They asked if I took any medications in the screening session, and I jokingly said 'well, I had a hay fever pill the other day'. They then asked, 'how many did you have?' and 'when did you take it?', so I assume they must have had a pretty rigorous screening set up.
What were you asked to do for the trial?
It was different for each participant group. I am in group 4. They gave us the vaccine, but they didn’t ask us to do an e-diary of the side effects because they’d already covered that in phase 1. I need to take a COVID-19 home test every Wednesday and send it off via priority mail.
Every Saturday I have to do an ‘exposure questionnaire’, asking questions such as; 'how many people have you seen within close contact this week?', 'how many times have you gone to a restaurant or a pub?', 'has anyone is your household tested positive?', and so on.
That’s surprising to hear that you didn’t have to record any symptoms.
No, my group didn’t have to. We were just asked to let them know if we reported anything severe. Although, they had pretty much managed the side effects by that point. They knew that it would cause aches and pains, maybe a bit of redness around the injection site, or a bit of swelling.
Therefore, none of that was unexpected. The placebo that I might have got (who knows?), the meningitis vaccine, also has quite a few of those side effects, even though it’s been licensed for a few years now. The researchers are thereby more interested about the efficacy of the vaccine, rather than its safety. They actually paused it one week because someone had an adverse reaction. They emailed us today to let us know what had happened. It seems a very standard part of the whole thing.
I need to take a COVID-19 home test every Wednesday
Did you have to get a booster shot?
Yeah, I got one now, because they believe a booster series to maintain the immune response is the solution. I will have another visit in 30 days, and then 60, and then 90. The results they have been getting, so far, suggests that there is a peak immune response at three and a half weeks post initial injection. Although they don’t know how long it actually stays effective and whether a booster would extend that exponentially.
While in Bristol this new round of vaccine recruitment is only now getting started, other COVID related studies have been ongoing. We interviewed Evy Tang, a fourth year computer science student at the University of Bristol, and a participant in an infection survey carried out in partnership with IQVIA, Oxford University and UK Biocentre COVID in Bristol.

What do the trials of this infection survey entail?
Someone with a medical background comes to our door and carries out some standard screening questions. These include questions such as if we’d been in contact with someone with COVID-19 in the last week, if we’d been social distancing, if we were wearing face masks when we were out, and if we’d seen any people under the age of 18. They seemed keen for kids in households to be part of the trial. We do the swab and then give it back.
When did you start the trial?
It started about four weeks ago. They take one weekly swab for the first month. After the first month, they reduce the swabs to once a month for an eleven-month period. So, a year length trial overall.
Could you describe the study you are currently part of?
We were sent a booklet about 2 months ago. Essentially Oxford University and the Office of National Statistics are trying to see how many people in England have already had the virus. They are doing this by recording how many people are infected within a sample group. Throughout this next year, the survey results can tell us where the infections tend to occur, and what kind of lifestyle the infected individuals lead.
Bristol researchers find a potential ‘game changer’ in the fight against COVID-19
Bristol researchers and the pursuit of a COVID-19 vaccine
How did you hear about the trials?
We were selected as a household. It seems that they based it on whether someone in the household had done something with the Office of National Statistics before. I haven’t heard of anyone else I know who has been selected, so it does not seem that common. I think now they have 1,000 people participating and they are looking to expand. We were just sent an information booklet directly to our address.
Were there any age ranges or other criteria they searched for that you are aware of?
No, that I am aware of, they just want as reflective of a population as possible.
Featured Image: Unsplash / Center for Disease Control
Are you currently looking to help out in COVID-19 research?