By Julia Riopelle, SciTech Editor
Research conducted by University of Bristol researchers has paved the way for a new treatment approach towards chronic kidney disease.
A Bristol-based gene therapy company, Purespring Therapeutics, has received £45 million in funding from their parent company Syncona Ltd, in order to develop a gene therapy based on the findings of Bristol Medical School. The clinical research has been pioneered by Professor Moin Saleem, a professor of Paediatric Renal Medicine and Dr Gavin Welsh, Associate Professor of Renal Medicine.
Syncona establishes, builds and funds companies in innovative areas of healthcare, science and medical research. They focus on investing in promising future treatments which can be rapidly delivered to patients.
Chronic kidney disease is a condition where kidney function is gradually lost over time. Amongst other problems, it causes fatigue, muscle cramping and frequent urination. Eventually it causes a build-up in toxic waste, which increases blood pressure, weakens bones, damages nerves and leads to anaemia. If left untreated, it leads to kidney failure and even death.
Kidney disorders are currently only treated via dialysis, where one is attached to a machine that takes over the role of the kidneys to filter the blood, or a kidney transplant. Due to these limited options there are currently only two million people who receive kidney transplants or dialysis worldwide, which is only around 10 percent of the people in need.
only two million people who receive kidney transplants or dialysis worldwide, which is only around 10 percent of the people in need
Thus, the cutting-edge research into renal gene therapy at the University of Bristol could allow for an alternative and more widely accessible treatment option. So far, Professor Saleem has been the only one to successfully use gene therapy to treat nephrotic syndrome in animals.
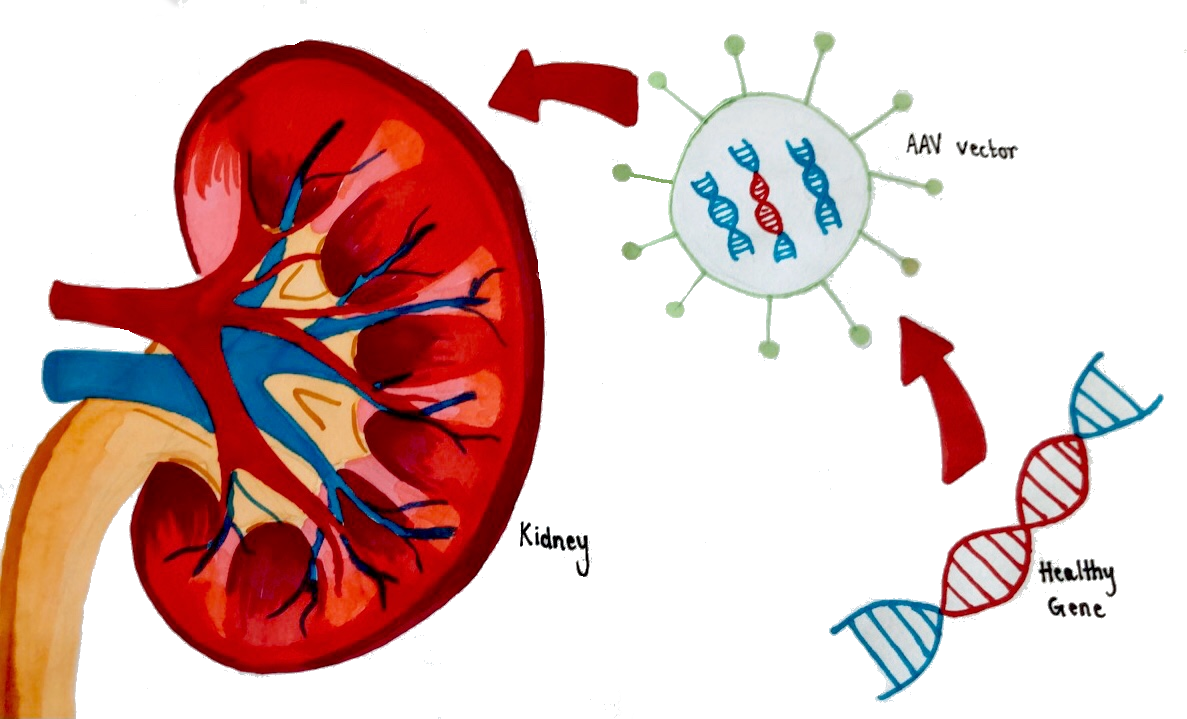
Gene therapy targets genes that cause an illness, either through replacing the faulty gene with a normal, healthy one, turning off or ‘silencing’ the genes causing the problems or by adding genes to help fight the illness. This type of therapy would not require drugs or surgery as part of the treatment plan.
Adding new genes to a cell requires a ‘vector’, a genetically engineered unit of DNA which can carry genes into the cell in order to be expressed. Viruses are commonly used as vectors, as they can transport the healthy gene to the desired tissues and are able to ‘infect’ host cells. They also have the ability to insert their DNA into their host’s DNA, in order for the cell to produce the proteins which the virus requires for survival. Scientists disable the virus’ ability to harm the host, in order to exploit this mechanism for gene therapy use.
The Platform Vector Gene Therapies Project: Increasing the Efficiency of Adeno-Associated Virus Gene Therapy Clinical Trial Startup https://t.co/pqbtKS1t4v#GeneTherapy #AAV @HGTJournal pic.twitter.com/nrkqHNAE1d
— Mary Ann Liebert Inc (@LiebertPub) November 24, 2020
Professor Saleem and Dr Gavin Welsh’s research uses Adeno-associated virus (AAV) gene therapy. AAV is ideal for this treatment because it does not cause human illness and is unable to replicate itself in the human body, as seen with other viruses. Thus, the patient’s body would not create an immune response to try to fight the ‘virus’ when introduced. AAV has also been observed to be able to insert its modified genes into the human chromosome 19 with 100 percent certainty.
The kidney’s role is vital to the human body, as it filters out waste, as well as regulates water and electrolyte levels in the blood. Too much water results in overhydration, which can lead to cell swelling and even brain swelling in extreme cases. However, dehydration causes electrolyte imbalances, hyperventilation, increases in heart rate and decreases blood pressure. The kidney also filters blood in order to remove drugs, toxins and urea.
The Purespring gene therapy will target the glomerulus, which is where the first stage of kidney filtration takes place. The glomerulus is a ‘knot’ of intertwined capillaries, located in a highly porous sac, which absorbs most of the waste from the blood into the kidney filtrate. The filtrate later leaves the body in the form of urine.
Bristol research reveals how cancer puts brakes on the immune system
Bristol-led research investigates treatment of mitochondrial diseases using gene editing
The funds granted to Purespring could rapidly speed up the time to get to the clinical trial phase of the renal gene therapy. If successful, they could be treating patients in just three to four years. Professor Moin Saleem commented, ‘This is an incredible opportunity to develop transformational treatments for kidney disease. Gene therapy has come of age in certain areas, but a major challenge in complex solid organs is to precisely target the genetic material to the correct cell type.’
Combining the expertise of these two companies with the expert knowledge of the Bristol research group, there is no doubt that huge progress is being made in the fight to cure chronic degenerative kidney conditions.
Featured Image: Epigram / Alice Proctor
Do you think gene therapies are the answer to untreatable diseases?