By Tom Farnell, Third Year Chemistry
Tom Farnell reports on exciting new developments in biosynthesis, an article published in Nature Chemistry by Bristol Researchers details the breakthrough.
Scientists at the University of Bristol have discovered that enzymes from the bacterium Pseudomonas fluorescensa play a key role in an important chemical reaction. The reaction involves the formation of a 6-membered ring of atoms from a long, chain-like molecule during the production of mupirocin, a medically important antibiotic. Mupirocin is used as a topical treatment for skin infections and as a pre-operative nasal spray to protect hospital patients from the multidrug-resistant bacteria MRSA, which is commonly found in hospitals. The ring formed during the reaction is known as a tetrahydropyran (THP) ring. THP’s and similar 5-membered rings are found in many biologically active natural products. The research, published in Nature Catalysis, could lead to cleaner and more efficient production of mupirocin and other antibiotics using new enzymes as biological catalysts to speed up the reactions. The ability to form these rings using biological catalysts would be particularly exciting, and could provide a potential route to increasing antibiotic production yields.
Professor Chris Willis from the School of Chemistry, who jointly led the study, called it an "important breakthrough”, and acknowledged that it “was achieved by an interdisciplinary team effort comprising talented postdoctoral researchers and postgraduates here at Bristol”. The process included “structural biology, synthetic and analytical chemistry in parallel with molecular modelling to unveil an overall transformation not previously reported for this family of enzymes."
Professor Matt Crump from Bristol’s School of Chemistry who was a supervisor on the study said that the work "serves as an example of the importance of nuclear magnetic resonance (NMR) spectroscopy in chemical and biochemical studies”, and notes that the access researchers had to the BrisSynBio-funded NMR spectrometer “enabled the identification of key intermediates in the pathway and opens future opportunities in synthetic biology which could not be achieved without the sensitivity of cutting-edge instrumentation."
The multidisciplinary team of researchers focused on cascade reactions in the formation of mupirocin. The ring formation step is essential in shaping the molecule correctly, so it can bind to bacteria like MRSA. The exact mechanism taking place in the reaction is not fully understood, although many mechanisms have been proposed. By monitoring the reaction while ‘turning off’ selected enzymes, the team discovered that the enzymes MupW and MupT are involved in ring formation. MupW is an oxygenase, meaning it is attracted to and binds to oxygen; MupT is a ferredoxin, meaning it is able transfer electronic charge. Further investigation of these enzymes revealed that MupW and MupT work together to create a chemical bond between an oxygen atom and two carbons in the starting material to produce an intermediate molecule called an epoxide. Another enzyme, MupZ was found to give the desired THP ring formation from the epoxide intermediate in the next step of the reaction cascade (image below). The selectivity of the MupW enzyme in ring formation is extremely valuable and a competitive route to those achieved through synthetic chemistry.
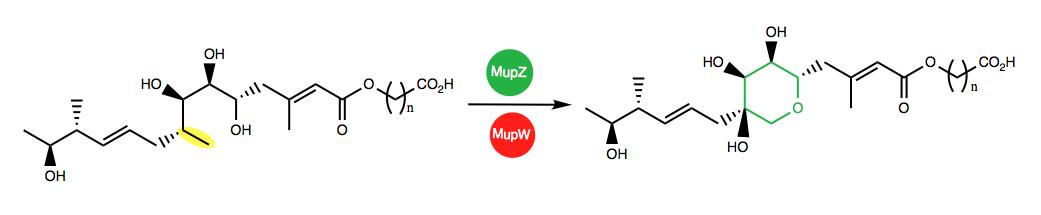
Image by Tom Farnell
The team is now investigating the enzymes involved to create improved versions of mupirocin with improved stability and stronger antibiotic effects to better treat MRSA infections and other illnesses. This research builds on previous studies by Bristol Professor Tom Simpson FRS and Professor Chris Thomas from the University of Birmingham. Dr Paul Race and Dr Marc van der Kamp from Bristol's School of Biochemistry were also part of the supervisory team carrying out the study at BrisSynBio, part of the Bristol Centre for Synthetic Biology, which was funded by the BBSRC and EPSRC.
Featured Image: Louis Reed/ Unsplash
What's your favourite piece of Bristol Research? Let us know!









